Homemade device for making living water
| Homemade device for making living water | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The plant has a multi-purpose purpose from its own healing and healing of wounds, to germination of seeds and acceleration of plant growth. In fact, it is noted that if the living water (anolyte) is watered in winter plants, then they begin to bloom. Ranks treated with dead water (catholyte) heal faster. In terms of improving my own body, I can say only one thing, this is purely individual and voluntary. Articles about the use of water broken into factions in its time was published very much - if you want them you can easily find. You can assemble this device without any preliminary preparation from completely improvised means found without leaving home. The main parts are electrodes. In the manufacture of special difficulties do not have. The only thing that must be noted - the material from which they are made must be only food grade stainless steel. Technical, as you know, for such an installation will not work. 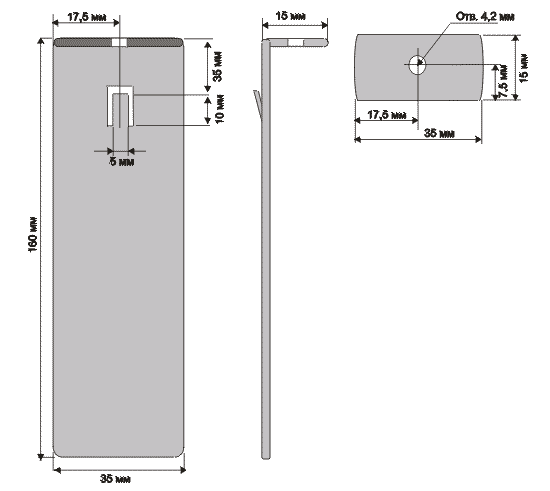 As can be seen from the figure - nothing complicated, two L-shaped platinum with a hole for fastening and contact. One plate has such a peculiar propyl - when bending at an angle of the middle protrusion, it is a hook for hanging the pouch. On the second plate, it is not necessary to make such a fixing propyl. The next detail for manufacturing is the stiffening plate. It is designed to avoid touching the electrodes in any way during operation of the device. 220 in the water, you know the thing is not pleasant. 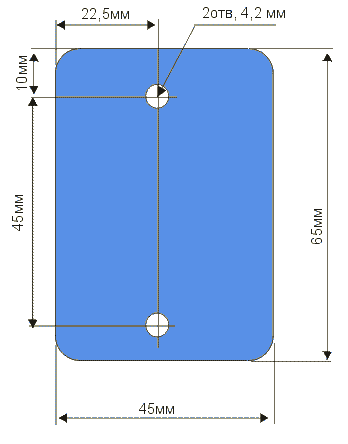 When using as a load-bearing structure of a conventional polyethylene lid for a jar, this plate will not be superfluous since there is little, if almost imperceptible, heating during the operation of the device, and how the behaved polyethylene behaves is known to you. So, just in case, it does not hurt. The next detail is a catholyte collection bag. It's just that it will be hard without women's hands. Material for the bag is a thin tarp. For example - I needed a tarpaulin from a gas bag. The criterion of selection is the heavy blowing of air through it. Synthetic materials are better not to use - what kind of muck is given in the work to guess difficult. When these components are ready, you can take the lid. The most common lid for a jar. In it, you need to make 3 holes: 2 fixing, one gas outlet. When using as a load-bearing structure of a conventional polyethylene lid for a jar, this plate will not be superfluous since there is little, if almost imperceptible, heating during the operation of the device, and how the behaved polyethylene behaves is known to you. So, just in case, it does not hurt. The next detail is a catholyte collection bag. It's just that it will be hard without women's hands. Material for the bag is a thin tarp. For example - I needed a tarpaulin from a gas bag. The criterion of selection is the heavy blowing of air through it. Synthetic materials are better not to use - what kind of muck is given in the work to guess difficult. When these components are ready, you can take the lid. The most common lid for a jar. In it, you need to make 3 holes: 2 fixing, one gas outlet. 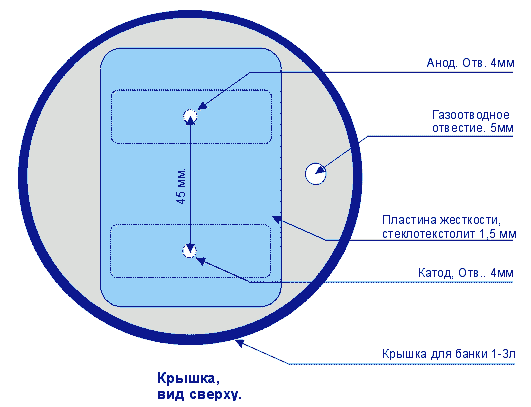 When the device is operating, gas is noticeably visible near the plates. Apparently, the treatment proceeds according to the type of weak hydrolysis, respectively, the release of gas - a by-product. This hole is just for leveling the pressure inside and outside the can with the lid closed. The order of assembly of the device is clearly visible in the figure below: 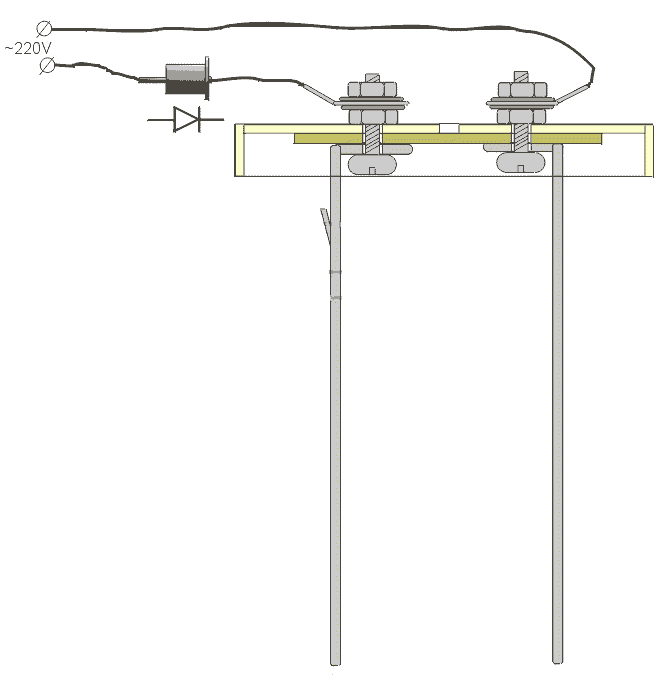 As you can see, nothing complicated. It can be noted that the nuts on each bolt must be two. One for fastening the electrode, the other for reliable contact. For the contact, the areas from the KD202 type diodes clamped between two washers are applied. As a diode, you can apply any with a working voltage of about 400 volts. The only condition is to connect it as it is shown in the figure, otherwise you will get catholyte not in the bag, but around it, i.e. All the way around. If you use a diode KD202, then attach directly to its thread directly anodic plate, note - anodic, because On the body of the anode. And the cathode was then connected directly to 220. True, I myself used a diode bridge in general. As you understand, the full-wave rectifier has twice the performance. On operation it is possible to notice the following: - The edges of the bag should be slightly above the water level, so that there is no mixing of the fractions. - The exposure time is 10-15 minutes. Or to lightly heat the jars. You can certainly raise literature and measure the acidity of catholyte litmus paper, etc., but it seems to me that it is superfluous. The fortress of catholyte can be determined by smell. - Do not use water directly from the tap. With chlorine contained in it, it is quite possible to assume that there will be HCl in the catholyte, i.e. hydrochloric acid. Water should be defended for at least 4-5 hours. All dimensions are based on a 3-liter jar Addition from our reader Mikhail Klychkov [email protected] Hello. The described method of obtaining "living" water has one drawback. The fact is that using the dividing bag is rather troublesome. To get "living" water should be very carefully, so as not to mix with "Dead". It is not comfortable. I came across an article with another way of getting "live" and "dead" water. The essence of the following. We need not one dielectric capacity for water, but two. Capacity needed Without necks; With straight walls. We put the containers next to a small gap and pour into both approximately An equal amount of water (but not to the brim). In one container we put the anode, In another cathode. Now a little fineness. It is necessary to make Conductor for installation between receptacles. To do this, pure cotton Roll is wrapped in a clean gauze and wetted with water. All this Is placed between the banks in such a way that the ends of the bundle are in both Banks, i. We connect the liquids of cans with such a peculiar bridge. On this jumper, ions will move and accumulate in different Capacity. After the "separation" of water, it will only be enough to turn off the voltage and Remove the "jumper" - "live" water in one bank, "dead" in the other. In the same article it is pointed out that in breaking one wire from Rectifier bridge in the electrical network need to include Electric bulb 15 ... 25 W to 220 V (such low power is apparently put in Refrigerators, lighting sewing machines, etc.). First, it will protect Circuit from a possible short circuit (at "k / z" in the circuit a light bulb Only something will burn in full heat). And secondly, it is needed as End-of-process indicator. We start to divide the water - the light is on. Gradually, the brightness of its glow decreases. When the lamp completely goes out, The separation of water can be considered complete. I hope this information will be useful.
M - dead water F - living water |


Comments
When commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.