Sulphides: Molybdenum
 Diagnostic card.
Diagnostic card.
In the photo: a sample from Arizona. Below: lamellar molybdenite on dolomite
Mo S 2
Shingoniya hexagonal
Hardness 1-1,5
Specific weight 4.6-5
Cleavage is perfect
Rough fracture
Color lead black
Color in powder greenish-gray
Glitter metal
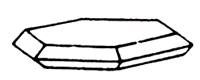
Molybdenum (molybdenum sheen) is molybdenum sulfide. The gloss is metallic, opaque. The color is lead-gray. The dash is also leaden-gray. Finely-cut has a greenish tinge. Flexible, but not elastic. Cleavage is very perfect. Genetically linked with granites, greisens, quartz veins, less frequently with pegmatites. Crystals (hexagonal system) are tabular. More often scaly, leafy or tabular aggregates. The most important ore of molybdenum. Deposits: in the USA, Canada, Chile, Norway, the CIS.
Molybdenite is represented by scaly plates of hexagonal outlines. Scales are often curved or twisted and have no clearly defined crystalline outlines. The mineral is opaque, the lead-gray color is bluish, with a bright metallic luster; It is heavy, often greasy to the touch, has perfect cleavage in the table. In thin plates flexible, but not elastic.
Chemical composition. Molybdenum (Mo) 60%, sulfur (S) 40%. Form of crystalline precipitates. Well-formed crystals are represented by hexagonal plates; In most cases molybdenite is represented by leafy and scaly aggregates and spherulites. Crystal structure. A typical layered lattice. Flat grids of the crystal lattice, formed by layers of molybdenum, are located between two layers of sulfur ions, parallel to the basal plane. Class of symmetry. Dihexagonally bipyramidal. Cleavage. Perfect in the basis of (0001). Aggregates. Thick, thin-scaly.



Molybdenite (lamellar crystals, 2 cm), on light topaz, with albite and yellow
Fine-grained pyrite. Volodarsk-Volynsky pegmatite, Ukraine, the CIS. 4,5х3,5 cm. Photo: © А.А. Evseev.
Diagnostic signs.
On a hexagonal form and very low hardness, molybdenite can be confused with graphite. A gray, pitted trait acquires an onion-green color (the graphite remains gray). They can be distinguished on the line on paper or on porous unglazed porcelain; Graphite gives a black-gray line, and molybdenite is greenish-gray. Does not melt. Behavior in acids. It is difficult to decompose in concentrated sulfuric acid with slow boiling.
Origin.
Pretty common and common mineral; Occurs mainly in high-temperature hydrothermal veins. Associated with minerals of tin, copper and tungsten. It is often found in granites and pegmatites.
Place of Birth.
In Italy, molybdenite is mined in many places. Good samples come from Arendal in Norway, Azegura in Morocco, Queensland in Australia and mainly from the areas of Temiskam and Oldfield Township in the province of Quebec (Canada). The Clymax field in the state of Colorado (USA) is developed in an industrial way.
Application.
Of molybdenite, molybdenum is recovered, used primarily for the production of special alloys. Due to its very low hardness and its ability to break down into tiny fatty particles, molybdenite finds application as a dry lubricant, especially at high temperatures.



Molybdenum acts as a cofactor in the enzymes responsible for detoxifying the body, affects the use of iron stores in the body, activates the exchange of sulfur-containing amino acids for the work of the nervous system and the brain (psychiatry), promotes retention of fluoride in the body, strengthening the enamel of the teeth.
Molybdenum is absorbed from food products and in the form of soluble complexes. 25-80% of molybdenum, which comes with food, is absorbed from people. Absorption occurs in the stomach and throughout the small intestine, more in its proximal part than in the distal ("corpse complex"). The absorption of molybdenum is affected by the interactions between molybdenum and various dietary (food) forms of sulfur (molybdenite).
About 80% of molybdenum, which enters the blood, binds to proteins (with albumin) and is transported through the body. In the blood, molybdenum is distributed evenly between the shaped elements and the plasma. Most of the molybdenum enters the kidneys and is excreted by them. Excretion is the main mechanism of its homeostatic regulation. Significant amounts are excreted with bile. The accumulation of molybdenum in the body does not occur (with the exception of the human brain).
Molybdenum is necessary: for neoplasms of the brain (tumor), visual impairment, tachycardia, male infertility. It is believed that tungsten, lead and sodium act as molybdenum antagonists and cause its deficiency in the body. Signs of insufficiency of molybdenum: increased excitability, irritability, impaired visual ("dark") adaptation, "night blindness," a violation of the rhythm of heartbeats (tachycardia), increased risk of esophageal cancer, decreased activity of enzymes that contain molybdenum.
Deficiency of molybdenum is possible in people who receive complete parenteral nutrition or are exposed to stress (increased need for sulfite oxidase). In patients receiving prolonged parenteral nutrition, the syndrome of "acquired deficiency of molybdenum" is described: hypermethioninemia, hypouricemia, hyperoxypurineemia, hypouricosuria and hyposulphaturia, progressive mental (cerebral and brain neural) disorders (to the coma - urgent conditions). Organs containing high amounts of molybdenum - liver and kidneys.
The main manifestations of excess molybdenum: increased activity of xanthine oxidase, an increase in uric acid in the urine; Gout (uraturia, urolithiasis); Irritation of mucous membranes, pneumoconiosis; Oppression hemopoiesis (anemia, leukopenia), weight loss. At excessive consumption - within 10-15 mg / day - the clinical symptoms of intoxication manifest themselves. At doses of molybdenum exceeding 15 mg / day, the activity of xanthine oxidase increases, uric acid accumulates, and the risk of gout increases (in persons in contact with molybdenum in production). When chronic molybdenum intoxication develops symptoms that are manifested by irritation of mucous membranes, pneumoconiosis, a decrease in body weight.
ADR 8
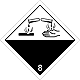
Corrosive (corrosive) substances
Risk of burns from skin corrosion. They can react violently with each other (components), with water and other substances. The substance that spilled / crumbled can emit a corrosive vapor.
Dangerous to aquatic environment or sewer system
White upper half of diamond, black - lower, equal, ADR number, test tubes, hands
| The name of a cargo that is particularly dangerous for transportation | room
UN |
Class
ADR |
| MOLYBDENA PENTACHLORIDE | 2508 | 8 |
- Ghetchellit - "New Almaden blend" - arsenide and antimony sulfide (modern sulfosol)
- Antimony is a toxic metal (semimetal) , widely used in metallurgy, medicine and engineering
- Zirconium - a rare and undiscovered metal and the most dangerous precious stone in oxide and salt
- Gold - yellow dangerous and poisonous metal of modern accurate digital and cable technologies
- Sulfur is a golden-yellow toxic substance and a sign of active volcanic activity
- Cadmium is an undisputed toxic silvery metal unknown to a wide range of people
- Lead - a toxic gray imitator of metallic silver and toxic metal blende
- Arsenic is a classic poison of medieval and modern poisoners and medicine in medicine
Poisonous and radioactive dangerous stones and minerals
** - poisonous stones and minerals (mandatory check in the chemical laboratory + explicit indication of toxicity)
** - radioactive stones and minerals (mandatory check on the standard dosimeter + ban on open sales in case of radioactivity exceeding 24 milli / g / h + additional measures of population protection)
Catalog of minerals and semi-precious stones of the world by groups
** - poisonous stones and minerals
** - radioactive stones and minerals


Comments
When commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.