Faldspathoids (silicates): Pollucite
 Diagnostic card.
Diagnostic card.
(Cs, Na) Al Si 2 O 6 * H 2 O
Cubic amount of cubic zirconia
Hardness 5-6
Specific weight 2.9
Cracked shell
Color is colorless, white, light gray
Color in powder white
Glitter glass
Pollucite crystallizes in the form of clear individuals, rich in facets, colorless, light gray or gray. Glitter is glass. Habitus is characterized by a combination of tetragon-trioctahedral and cube. It often forms dense irregular aggregates or small veins, where it is partially replaced by kaolin.
Diagnostic signs.
It melts easily, giving a light gray glass and coloring the flame with a yellowish-pinkish color. The action of acids almost or completely does not lend itself.
Origin.
Pollucite is a mineral of hydrothermal genesis, very rare in nature. Its typical location is granite pegmatites, where it is usually found together with other minerals.
Deposits and applications.
It was developed in the middle of the XIX century, in the form of drusen with petalite in the granites of San Pietro in Campo on the island of Elba. Rich concentration is in Verrecesk in Sweden, in Karibib in Namibia, in the spodumene deposit in Bernick Lake (USA). Pollucite is the main ore for cesium.

Pollucite. Kulam, Afghanistan. Crystal ~ 10 cm. Photo: © А.А. Evseev.

Pollucite. Area Gilgit - Skardu, top. R. Indus, Pakistan. Tucson-show-2007.
Cesium is relatively low in toxicity, its biological role in the human body is not fully understood, although it is assumed that it plays a role in the preservation of human homeostasis, it is effective at a sharp decrease in blood pressure (syncope, shock states, collapse) - similar to potassium in the body and has a property Accumulate, replacing this element. The daily requirement of the human body is not established.
Getting into the body with food, cesium (like potassium, the main element) is quickly absorbed from the gastrointestinal tract into the bloodstream. Cesium is evenly distributed in organs and tissues, mainly enters the intestine and is reabsorbed again in its descending sections. About 80% of the cesium that enters the body accumulates in the muscles, 8% in the skeleton, the remaining 12% are distributed evenly over other organs and tissues (heart, liver, up to 2.8 μg / l in the blood). In the human body 137-Cs is distributed relatively evenly and does not have a significant deleterious effect. Cesium is excreted mainly through the kidneys and intestines (after replacing it with potassium ions).
Biological role in the human body. The role of cesium in certain physiological processes is known. The stimulating effect of this element on blood circulation functions and the effectiveness of its salts application in hypotension of various origin have been established. Proceeding from the expressed hypertensive and vasoconstrictive action, the salts of cesium as early as 1888 were first applied by a Russian physician, who worked in the laboratory of I.P. Pavlova, - S.S. Botkin - with violations of the cardiovascular system. He was found that cesium chloride causes an increase in blood pressure for a long time, and this effect is mainly due to increased cardiovascular activity and narrowing of peripheral vessels.
Cesium, enhancing and prolonging the action of endogenous vasomotor drugs, has a positive effect in hypotension of various origins. Cesium salts in optimal doses promote the rapid restoration of a catastrophically lowered arterial pressure for various types of shock and collapse (with their subsequent excretion from the body and the replacement of potassium salts). The adrenomimetic and sympathomimetic effects of cesium salts on central and peripheral adrenoreactive structures have been established, which is especially pronounced in the suppression of the tone of the sympathetic part of the central nervous system and the deficit of catecholamines. Cesium salts are characterized, mainly, by the? -adrenostimulating effect.
Cesium salts affect nonspecific indices of immunobiological resistance - they cause a significant increase in the complement titer, lysozyme activity, and phagocytic activity of leukocytes. There are indications of the stimulating effect of cesium salts on the functions of the hematopoietic organs. In micro doses they cause stimulation of erythro- and leukopoiesis (by 20-25%), markedly increase the resistance of erythrocytes, increase the hemoglobin content in them. Cesium chloride participates in gas metabolism, activating the activity of oxidative enzymes, cesium salts increase the body's resistance to hypoxia.
Synergists and antagonists of cesium. Cesium synergist is rubidium. Signs of cesium deficiency: decreased appetite; Stunted growth and development. Cesium is necessary (as a temporary replacement of potassium in the body): in shock states, fainting, collapse, hypotension, ulcerous diseases, diphtheria. Food sources of cesium: the accumulation of cesium is maximized in the tissues of freshwater algae and arctic terrestrial plants, especially lichens. A high content of cesium is observed in leaf lettuce (in roots) and in mushrooms of honey mushrooms. In the animal body, cesium accumulates mainly in the muscles and liver. The highest accumulation coefficient was observed in reindeer and northern American waterfowl. On the materials: http://www.pharmacognosy.com.ua
Cesium-137. In the case of long-term radioactive contamination, preventive measures are to control food chains. First of all, it concerns broad-leaved plants (spinach, lettuce, burdock - medicinal plant, roots) and milk (due to consumption of grass for food by ruminant animals). Among all radioactive isotopes of cesium, the first place belongs to cesium-137. Its half-life is 30 years, which is much higher than that of iodine-131. It is an extremely toxic and radioactive substance that often settles in plants and mushrooms.
That is why decades later after the accident in Chernobyl (Chernobyl, Chernobyl, Ukraine, CIS), boars were found in the Vosges (and they, by the way, are big fans of eating mushrooms) with an increased level of radiation in the body. Although from the point of view of cesium chemistry is rather close to potassium (this element is an integral part of the processes of cellular metabolism in the body) there is practically no way to get rid of it. Cesium can cause cancer of the digestive system, lungs or blood (leukemia). The last cleavage product, strontium-90, has an even more significant half-life, is close in its chemical properties to calcium in our body and can be found in milk during the food chain. On the materials: http://inosmi.ru/fareast/20110329/167865529.html
ADR 4.3


Substances that emit flammable gases in contact with water
Risk of fire and explosion if exposed to water.
The cargo, which crumbled, must be covered and kept dry
Blue and blue diamond, ADR number, black or white flame
ADR 5.1
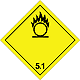
Substances that are oxidized
Risk of violent reaction, ignition or explosion if exposed to flammable or flammable substances
Do not allow the formation of a mixture of cargo with flammable or combustible substances (eg sawdust)
Yellow diamond, ADR number, black flame above the circle
ADR 8
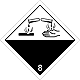
Corrosive (corrosive) substances
Risk of burns from skin corrosion. They can react violently with each other (components), with water and other substances. The substance that spilled / crumbled can emit a corrosive vapor.
Dangerous to aquatic environment or sewer system
White upper half of diamond, black - lower, equal, ADR number, test tubes, hands
| The name of a cargo that is particularly dangerous for transportation | room
UN |
Class
ADR |
| CESIUM | 1407 | 4.3 |
| CESIUM HYDROXIDE | 2682 | 8 |
| CYCLE OF HYDROXIDE SOLUTION | 2681 | 8 |
| CESIUM NITRATE | 1451 | 5.1. |
- Ghetchellit - "New Almaden blend" - arsenide and antimony sulfide (modern sulfosol)
- Antimony is a toxic metal (semimetal) , widely used in metallurgy, medicine and engineering
- Zirconium - a rare and undiscovered metal and the most dangerous precious stone in oxide and salt
- Gold - yellow dangerous and poisonous metal of modern accurate digital and cable technologies
- Sulfur is a golden-yellow toxic substance and a sign of active volcanic activity
- Cadmium is an undisputed toxic silvery metal unknown to a wide range of people
- Lead - a poisonous gray imitator of metallic silver and toxic metal blende
- Arsenic is a classic poison of medieval and modern poisoners and medicine in medicine
Poisonous and radioactive dangerous stones and minerals
** - poisonous stones and minerals (mandatory check in the chemical laboratory + explicit indication of toxicity)
** - radioactive stones and minerals (mandatory check on the standard dosimeter + ban on open sales in case of radioactivity exceeding 24 milli / g / h + additional measures of population protection)
Catalog of minerals and semi-precious stones of the world by groups
** - poisonous stones and minerals
** - radioactive stones and minerals


Comments
Commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.