Native elements: Antimony
 Diagnostic card.
Diagnostic card.
Sb
The trigonial trigonometric system
Hardness 3-3,5
Specific weight 6,7
Cleavage is perfect
Crack irregular
Color white silver
Color in powder gray
Glitter metal
Occasionally observed in the form of individuals pseudocubic or tabular appearance. The most common form is granular masses, incrustations and contractions of the radiant structure. The color is white silver, the shine is metallic, very strong.
Sometimes it contains Ag, Fe or As. Solid granular secretions, less often infiltrating aggregates (kidney-shaped, grove-shaped), sometimes radiant structure; Crystals are rare. Structure and morph. Crist. Trig. from. D53d-R3m; Arh = 4.507 A; A = 57 °o 06; Z = 2; Ah = 4,310; Ch = 11.318 A; Ah: ch = 1: 2,627; Z = 6. Structure of the arsenic type. The distances of Sb-Sb are 2.87 and 3.37 A. The Dietrigon-skalenohedron. Class; A: с = 1: 1,3236 Crystals rhombohedral, thick-plate (0001) or plate-like. Two. By (1012); Form complex groups - quadrants, gears, often polysynthetic. Cleavage according to (0001) is perfect, according to (2021) it is sometimes clear, according to (1120) and according to (1012) imperfect.
Diagnostic signs.
Low hardness (it is scratched by a penknife). In acids it does not dissolve, it burns in the air, emitting white smoke. Antimony (English Antimony, French Antimoine, German Antimon) man knows long ago in the form of metal, and in the form of some compounds.
Origin.
Antimony is usually of hydrothermal origin. Formed during the solidification of residual fluids in the process of crystallization of granites and pegmatites. Therefore, it occurs in hydrothermal veins together with complex sulfoarsenates and sulfoantimonates, mainly of metals such as silver.
Deposits and applications.
Small amounts of this mineral are found in Sarawak, Borneo, Sala (Sweden), Andreasberg (Germany), Coimbra (Portugal), Canada and California. For industrial use, antimony is extracted from its salts. Native metal is only a scientific and a collector's interest.

Antimony. Dauphine, France. Photo: © А.А. Evseev.


Antimony. Seinäjoki, Finland. Antimony. Burkandiya, Magadan region, Russia (SW). Photo: © А.А. Evseev.

Antimony is massive and fine-grained. Burkandiya, Susumana district, Magadan region, Russia, CIS. Photo: © А.А. Evseev.
Antimony - on the slang "vomiting stone." Antimony enters the body with food and selectively concentrates in the thyroid gland, liver, spleen, skeleton, kidneys, blood (in erythrocytes) and in other soft organs and human tissues. In erythrocytes, antimony accumulates in the oxidation state +3, in the blood plasma (lymphocytes) - in the degree of oxidation +5. From the body, antimony is excreted slowly, with urine (up to 80%).
The maximum permissible concentration of antimony in the human body is 10-5-10-7 g per 100 g of dry tissue mass. At high concentrations, antimony inactivates a number of enzymes of lipid, carbohydrate and protein metabolism (as a result of blocking of sulfhydryl groups, replaces red cinnabar - dry red mercury sulphide).
When taking antimony drugs inside in toxic doses, acute or chronic poisoning can develop. Antimony forms bonds with sulfur atoms (reacts with sulfhydryl (thiol) groups of enzymes), which causes its high toxicity (antimonite). The main manifestations of excess antimony: loss of appetite, inflammation of the mucous membranes of throat and larynx, dryness in the throat, nausea, vomiting, pain in the intestine, an increase and soreness of the liver, icteric sclera; A prolonged cough.
The general effect of antimony preparations is similar to the action of arsenic. Antimony has a more abrupt local effect, causing severe vomiting of reflex origin, severe anatomical lesions of the gastrointestinal tract; Antimony preparations are more difficult to absorb, and the poisoning proceeds more slowly. With the use of antimony preparations, fatty degeneration and cirrhosis of parenchymal organs, liver and kidneys develop. Significant toxicity limited the use of antimony preparations for medical purposes as self-medication (brain psychiatry).
For a long time, antimony compounds, such as antimony pentasulphide, wine-arsenic potassium ("vomitic stone") and wine-surmyanosodium salt were used as expectorants and emetics. To induce vomiting, the patient was given wine, aged in an antimony vessel. And, in addition, antimony replaces red cinnabar (poisonous mercury sulfide) in the treatment of psychiatric disorders and hallucinations - under electric shock (electrodes in the brain). In the early twentieth century. Antimony compounds were also used as expectorants and antiparasitic agents.
Antimony oxide (III) in ancient Greece served to treat skin diseases, and in the Middle Ages was used as a therapy for leprosy, syphilis and heart diseases. In modern medicine, antimony preparations (solyusurmine, etc.) are used to treat a number of infectious diseases of humans and animals (brain pathologies, like red cinnabar - mercury sulphide, replace each other). They are used in the treatment of visceral and cutaneous leishmaniasis, African trypanosomiasis (sleeping sickness), in the study of blood coagulation (red cinnabar, in the complex of electroconvulsive therapy). Food sources of antimony: seafood, vegetables, fruits, juices, drinks.


Antimonitis. Shikoku Island, Japan. Brush of pinacoid prismatic crystals. Baia-Spree, Romania. Photo: © А.А. Evseev.
ADR 6.1
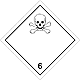
Toxic substances (poison)
Risk of poisoning by inhalation, in contact with skin or if swallowed. Dangerous to aquatic environment or sewer system
Use a mask for emergency leaving the vehicle
White diamond, ADR number, black skull and crossbones
ADR 8
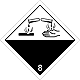
Corrosive (corrosive) substances
Risk of burns from skin corrosion. They can react violently with each other (components), with water and other substances. The substance that spilled / crumbled can emit a corrosive vapor.
Dangerous to aquatic environment or sewer system
White upper half of diamond, black - lower, equal, ADR number, test tubes, hands
| The name of a cargo that is particularly dangerous for transportation | room
UN |
Class
ADR |
| SB - POWDER | 2871 | 6.1. |
| Antimony Pentaphthyride Antimony Pentaphthoride | 1732 | 8 |
| Antimony Lactate | 1550 | 6.1. |
| Antimony Pentafluoride | 1732 | 8 |
| Antimony PENTACHLORIDE LIQUID | 1730 | 8 |
| Antimony PENTACHLORIDE SOLUTION | 1731 | 8 |
| SBAN COMPOUND INORGANIC LIQUID, NZK | 3141 | 6.1. |
| SBM INORGANIC COMPOUND, SOLID, NZK | 1549 | 6.1. |
| ANTIMONY TRICHLORIDE SOLID | 1733 | 8 |
| Antimony Potassium Tartrate | 1551 | 6.1. |
Poisonous and radioactive dangerous stones and minerals
** - poisonous stones and minerals (mandatory check in the chemical laboratory + explicit indication of toxicity)
** - radioactive stones and minerals (mandatory check on the standard dosimeter + ban on open sales in case of radioactivity exceeding 24 milli / g / h + additional measures of population protection)
Catalog of minerals and semi-precious stones of the world by groups
** - poisonous stones and minerals
** - radioactive stones and minerals


Comments
When commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.