| Start of section
Production, amateur Radio amateurs Aircraft model, rocket-model Useful, entertaining |
Stealth Master
Electronics Physics Technologies Inventions |
Secrets of the cosmos
Secrets of the Earth Secrets of the Ocean Tricks Map of section |
|
| Use of the site materials is allowed subject to the link (for websites - hyperlinks) | |||
SELF-MODULAR PLANT FOR GALVANIC COATING
METAL PRODUCTS
![]()
See also: |
In the repair and amateur practice, it is also possible to successfully use a miniature empty-free galvanic installation (Figure 1). It consists of a special brush with bristles (brush diameter 20-25 mm , its body is made of organic glass, inside of which an electrolyte is poured), a step-down transformer for 12 volts In addition, the current is 0.8-1 A or the battery is also a connecting cord.
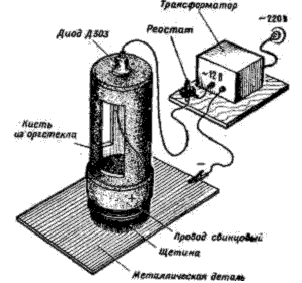
Fig. 1. Miniature galvanic installation
Bristle bristles are wound with lead (in extreme cases irradiated copper) wire. The semiconductor diode D303-D305 is installed on the body of the brush. One of the wires of the winding of the transformer is connected to the anode of the diode, the other by means of the clamp "crocodile" - with the part to be coated. The diode cathode is connected to the winding wire of the brush. If a battery is used, the diode is not needed.
The parts to be covered are cleaned of rust, grease is also grease, wiping with a dry clean cloth is also degreased in a solution containing 100-150 g of sodium hydroxide, 40-50 g of soda ash plus 3-5 g of liquid glass (silicate adhesive) per 1 l . Depending on the degree of contamination, the parts are kept in a degreasing composition, heated to 80-100 ° C , from 15 minutes up to 1 hour . The smoother the surface is, the more durable will be the galvanic coating. The prepared part is connected with the winding of the transformer, the brush is poured into the electrolyte and also includes feeding. Evenly moving the brush on the surface of the part, cover it with metal, deposited from the electrolyte. To obtain a coating of sufficient thickness, it is necessary to brush by the same same room 20-25 times. Electrolyte is added to the brush by the measure of its consumption. After coating, the part is washed in running water and also polished in a cloth moistened with water, then again washed and also dried. For each type of coating, a special electrolyte is prepared, prepared according to the following recipes (in grams per liter of solution):
|
Electrolyte for copper plating:
|
Electrolyte for galvanizing:
|
Electrolyte for nickel-plating:
|
Electrolyte for silvering:
|
|
Electrolyte for chrome plating:
|
Electrolyte for gilding:
In 200-300 ml of distilled water, dissolve the first order of the substance, then the second, third, etc., only then add water to 1 liter . It should be borne in mind that, although solutions also do not contain much poisonous substances, they should be handled with care to avoid burns and poisoning. Solutions are best preserved in a dark glass container with ground glass stopper. It is necessary to take into account that not all coatings fit well on various metals. For example, in order to coat a steel part with nickel, it is preliminarily coated with a thin layer of copper, chromium is well adhered to the nickel-plated surface. A copper sublayer before nickel plating or silvering is desirable to be deposited on parts made of bronze. Copper also brass parts silver without a sub-layer of copper.  Liked? Subscribe to RSS news!
You can also support shram.kiev.ua, press: It will not be superfluous for your friends to learn this information, share their article with them! |



Comments
When commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.