| Start of section
Production, amateur Radio amateurs Aircraft model, rocket-model Useful, entertaining |
Stealth Master
Electronics Physics Technologies Inventions |
Secrets of the cosmos
Secrets of the Earth Secrets of the Ocean Tricks Map of section |
|
| Use of the site materials is allowed subject to the link (for websites - hyperlinks) | |||
SIMPLE CASE APPARATUS
CO 2 SUPPLY IN AQUARIUM
Vadim Lisovsky
![]()
Glossary of terms |
|
DESIGN
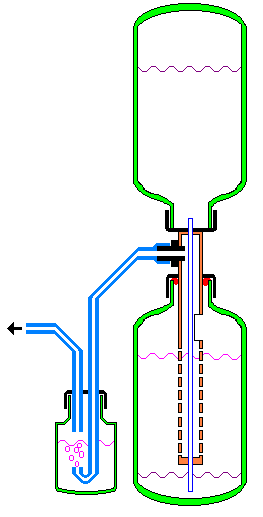
The device consists of a pair of identical bottles of PEF ( green color ), also a plastic tube ( orange ), the diameter of the bottle entering the throat. The tube is closed with plastic stoppers on a pair of sides, the upper plug in the figure must ensure tightness. In the upper elements of the tube, not in any way included in the bottom bottle, there is a nesting piece for CO2 removal ( black color ). Below it is also put on the cork from the bottle ( black color ), the tightness of this collapsible joint provides a rubber or silicone ring ( red color ). Under the cork made a spacious cutout for easy loading of chalk or limestone. Still further, the tube is perforated by a plurality of holes for free acid contact, also chalk. Through the top also the bottom stoppers the polyethylene tube for a cocktail ( blue color ) is punched. It is hermetically sealed also in the cork of the top bottle - it is best to insert a plastic hollow rod inside its upper end, which securely jams the tube in the opening of the cork of the upper bottle. In order to neutralize acid vapors in the waste gas, it is bubbled through a solution of soda in a separate bottle.
|
OPERATING PRINCIPLE The production of CO 2 is based on the well-known reaction between chalk and acetic acid. Chalk or limestone is needed in the form of pieces that do not pass through the opening of the tube, and especially there should not be any chalk dust. The device is filled with a weak solution of acetic acid almost up to the upper level. Shown in the figure. In the tube of the upper elements of the apparatus is placed pieces of chalk, it is inserted into the bottle, and the release of carbon dioxide also begins. It is drained through the union, it breaks through a solution of soda and is also fed into the aquarium. On the output is very desirable to put a non-return valve. If the release of gas is not extremely violent - the apparatus works as if just a container with chalk is also acid. But if CO 2 is released more than necessary, or the CO 2 outlet is blocked (by a tap or valve), the evolved gas starts to force out the liquid from the lower bottle and it also ascends the top bottle along the central tube. The reaction of gas evolution ceases, at what time the level of acid in the bottom bottle is made below the perforated chalk tube and also the basic capacity of the acid passes into the upper bottle. If the gas withdrawal is resumed, the pressure in the lower bottle drops as well, the acid again starts to react with chalk. Thus, according to the principle of the Kipp apparatus, a constant gas pressure is maintained in certain limits. This pressure, respectively, the waste of gas, will also depend on the depth of immersion of the nebulizer in the aquarium. |
REAGENTS
The rapidity of the reaction depends strongly on the content of calcium carbonate in the used chalk or limestone, the density of the porosity of the mineral. I advise you to first choose the concentration of acid on a small sample - the evolution of the gas should be sufficiently weak, unacceptably strong foaming, as well as bubbling. If the slow course of the reaction does not provide the right amount of gas in any way, it is better to re-assemble the device by screwing the bottles of a larger capacity than using a more concentrated acid. On the other hand, a small concentration of acid can lead to the need to more often transfer the device - it will become extremely neutralized. In general, it will be necessary to select.
FOR WHAT IS IT ALL ABOUT
As I present, there are some benefits - CO 2 costs less than from brags also from cylinders - because the device itself is not worthless either, the reagents are cheap enough (limestone is general free). The device at the expense of its device allows without a special risk to stop the flow of CO 2 at night - the release of gas automatically stops. Disadvantages - will have to work hard to reliably also hermetically assemble all of this, which is decently harder than withdrawing from a bottle with a brag. But you can also buy in the shop of chemical devices also finished such a device of glass  Just do not forget also the external grid for it - such a device with hydrogen decently spread on my eyes also flying in all directions splinters of glass - in no way the most pleasant thing.
Just do not forget also the external grid for it - such a device with hydrogen decently spread on my eyes also flying in all directions splinters of glass - in no way the most pleasant thing.
ATTENTION !!!
This device is working under small pressure, but also with a weak but acid.
If you are not sure of the strength of the connections made by the elements of the device - put it entirely
In a large capacity or a strong leakproof plastic bag!
Author of the article: Vadim Lisovsky




Comments
When commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.