Sulphates: Epsomite
 Diagnostic card.
Diagnostic card.
Mg SO 4 * 7 (H 2 O)
Singonia monoclinic
Hardness 2-2,5
Specific weight 1,7
Cleavage is perfect
White color
Color in powder white
Glitter glass
Epsomite occurs in the form of fibrous and stalactite masses, efflorescences and crusts with a fibrous structure. The color is white, the shine is glassy to silky, and in the differences between the fibrous and continuous structure - to earthy. Chemical composition. Magnesium oxide (MgO) 16.3%, sulfate (SO3) 25.5%, water (H2O) 51.2%; As an isomorphous impurity, ferrous oxide (FeO) (ferruginous epsomite) or nickel oxide (NiO) (nickel-epsomite) can be present.
Class of symmetry. Rhombo-dysphenoidal - 222. Form of crystalline secretions. Blots, incrustations (icicles), granular, fibrous, earthy masses, sometimes crystals up to 10 cm in size. Cleavage. Good for (011); The mineral is very fragile.
Diagnostic signs.
Easily soluble in water. The taste is bitter ("bitter", or "English, salt"). Due to the content of nickel or cobalt impurities, the color may be greenish or pink. When heated, it forms a non-melting white mass, at a temperature of 100-238 o C loses crystallization water.
Origin.
It is formed mainly on deposits of table salt, and also in the form of felt-like efflorescences in the regions of development of limestone and dolomite sequences. In addition, it can be deposited from thermal springs and formed in the oxidation zone of pyrite deposits.
Place of Birth.
The typical known long-standing location of the epsommit is Epsom in Surrey county in England, where this mineral is deposited from the waters of thermal springs. Other deposits - Sedlice in Bohemia (Czech Republic); Stassfurt in Saxony (Germany); Kalatyud in Spain; In the United States (Oroville, Washington), in the Sydney area (Australia). In Italy, there were reports of findings of epsomite in Valle Antron.
Application.
Epsomite is used in many industries: paper, pharmaceutical, tanning (as a mordant) and textile (for dyeing fabrics).
Magnesium - a component of enzymes, in the human body is contained in bones, teeth, is the regulator of the work of the nervous system. Magnesium is a macronutrient, from the insufficiency of which up to 90% of people suffer. In the human body contains about 140 g of magnesium (0.2% of body weight), with 2/3 accounted for bone tissue. The main "depot" of magnesium is found in bones and muscles in the form of phosphates and bicarbonates. Magnesium enters the body with food (in particular, with table salt - bitter, "Artemsol") and water.
Magnesium depletion is noted in rickets. Signs of magnesium deficiency: worsening of the transmission of nervous and muscle impulses, causing irritability and nervousness, increased excitability, spasms and convulsions, disorientation in time and space, insomnia, migraine, chronic fatigue, digestive disorders, palpitations, attacks, outbursts of anger or irritation. The first sign of magnesium deficiency are twitching and convulsions, especially the calf muscles. Psychiatric mineral.
Magnesium deficiency is the basis of cardiovascular disease. Against the background of a permanent magnesium deficiency, the risk of atherosclerosis of the arteries of the heart, heart attacks and changes in the rhythm of the heart increases. It can be the cause of lethal cardiac arrhythmia, hypertension, sudden cardiac arrest, asthma (especially on the basis of inhaling cinnabar), chronic pain syndrome (posttraumatic syndrome), depression, insomnia, irritable bowel syndrome and lung diseases. Arthritis and osteoporosis develop. Most magnesium is found in wheat bran and cereal sprouts (520 and 270 mg per 100 g).
Magnesium is necessary: for hypertension, symptomatic hypertension, atherosclerosis, liver and bile duct disease, depression, dizziness, muscle weakness and convulsive contractions, psoriasis, scleroderma, lupus erythematosus, premenstrual syndrome, to maintain a normal pH balance in the body, to prevent calcification of soft Tissues. It protects the endothelium of arteries from stress caused by sudden changes in blood pressure, it is necessary for the formation of bone tissue, carbohydrate and mineral metabolism, for dissolution of kidney stones formed from oxalates and phosphates. Magnesium enhances the effectiveness of the action of vitamin B6 (pyridoxine), important for the prevention of atherosclerosis.
An increase in magnesium (hypermagnesia) in the blood can occur with the intake of antacids or laxatives containing magnesium in patients with chronic renal insufficiency. When parenteral administration of magnesium sulfate symptoms of intoxication in the form of general oppression, lethargy and drowsiness (the brain is involved), heart failure can be observed. The use of a solution of magnesium sulphate during pregnancy and childbirth 4 times increases the risk of developing cerebral palsy in newborns (weak mothers). Anesthesia (before coma) occurs when the concentration of magnesium in the blood is 15-18 mg%.
Magnesium is even afraid of doctors, it is a dangerous "comatose" ("sleepy") drug that depresses the brain - almost 100% hospital (when treated with medical magnesia). The mineral, whose ions "work" at the junction of neurons (axons). In acute renal failure with oliguria, especially in combination with metabolic acidosis, hypermagnesia is possible. Infusion of calcium (an antagonist) can counteract the toxicity of magnesium.
ADR 4.1

Highly flammable solids , self-reactive substances and solid desensitized explosives
Risk of fire. Flammable or combustible substances can ignite from sparks or flames. May contain self-reactive substances capable of exothermic decomposition in the case of heating, contact with other substances (such as: acids, heavy metal compounds or amines), friction or impact.
This can lead to the emission of harmful or flammable gases or vapor or spontaneous combustion. Containers can explode when heated (over-dangerous - practically do not burn).
Risk of explosion of desensitized explosives after loss of desensitizer
Seven vertical red stripes on a white background, equal in number, ADR number, black flame
ADR 4.2

Substances that are capable of self-ignition
Risk of fire due to autoignition in the event that the packaging is damaged or the source of the contents has occurred.
Can react violently with water
White upper half of diamond, red - lower, equal, ADR number, black flame
ADR 4.3


Substances that emit flammable gases in contact with water
Risk of fire and explosion if exposed to water.
The cargo, which crumbled, must be covered and kept dry
Blue and blue diamond, ADR number, black or white flame
ADR 5.1
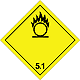
Substances that are oxidized
Risk of violent reaction, ignition or explosion if exposed to flammable or flammable substances
Do not allow the formation of a mixture of cargo with flammable or combustible substances (eg sawdust)
Yellow diamond, ADR number, black flame above the circle
ADR 6.1
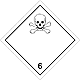
Toxic substances (poison)
Risk of poisoning by inhalation, in contact with skin or if swallowed. Dangerous to aquatic environment or sewer system
Use a mask for emergency leaving the vehicle
White diamond, ADR number, black skull and crossbones
| The name of a cargo that is particularly dangerous for transportation | room
UN |
Class
ADR |
| Magnesium nitrate see MAGNESIUM NITRATE | 1474 | 5.1. |
| MAGNESIUM - POWDER or MAGNESIUM OF ALLOYS - POWDER | 1418 | 4.3 |
| MAGNESIUM or MAGNESIUM ALLOYS, which contain more than 50% magnesium (granules, chips or tapes) | 1869 | 4.1 |
| Magnesium silicate fluorides, see MAGNESIUM FLUOROSILICATE | 2853 | 6.1. |
| MAGNESIUM IN GRANULES COATED, particle size not less than 149 microns | 2950 | 4.3 |
| Magnesium phosphorous (Mg3 (PO3) 2) see Magnesium phosphide (Mg3P2) | 2011 | 4.3 |
| MAGNESIUM DIAMID | 2004 | 4.2 |
| MAGNESIA ARSENAT | 1622 | 6.1. |
| MAGNESIUM BROMATE | 1473 | 5.1. |
| MAGNESIUM HYDRID | 2010 | 4.3 |
| Magnesium methyl bromide in ether diethyl see METHYLMAGNEBROMIDE IN ETHYL ETHER | 1928 | 4.3 |
| MAGNESIUM NITRATE | 1474 | 5.1. |
| MAGNESIUM PEROXIDE (peroxide) | 1476 | 5.1. |
| MAGNESIA PERHLLORAT | 1475 | 5.1. |
| Magnesium silicate fluoride see MAGNESIUM FLUOROSILICATE | 2853 | 6.1. |
| MAGNESIUM SILICIDE | 2624 | 4.3 |
| MAGNESIUM PHOSPHIDE | 2011 | 4.3 |
| MAGNESIUM FLUOROSILICATE | 2853 | 6.1. |
| MAGNESIUM CHLORATES | 2723 | 5.1. |
| Magnesium chloride, solution | - | - |
| MAGNESIUM-ALUMINUM PHOSPHIDE | 1419 | 4.3 |
- Ghetchellit - "New Almaden blend" - arsenide and antimony sulfide (modern sulfosol)
- Antimony is a toxic metal (semimetal) , widely used in metallurgy, medicine and engineering
- Zirconium - a rare and undiscovered metal and the most dangerous precious stone in oxide and salt
- Gold - yellow dangerous and poisonous metal of modern accurate digital and cable technologies
- Sulfur is a golden-yellow toxic substance and a sign of active volcanic activity
- Cadmium is an undisputed toxic silvery metal unknown to a wide range of people
- Lead - a toxic gray imitator of metallic silver and toxic metal blende
- Arsenic is a classic poison of medieval and modern poisoners and medicine in medicine
Poisonous and radioactive dangerous stones and minerals
** - poisonous stones and minerals (mandatory check in the chemical laboratory + explicit indication of toxicity)
** - radioactive stones and minerals (mandatory check on the standard dosimeter + ban on open sales in case of radioactivity exceeding 24 milli / g / h + additional measures of population protection)
Catalog of minerals and semi-precious stones of the world by groups
** - poisonous stones and minerals
** - radioactive stones and minerals


Comments
When commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.