Sulphides: Galena
 Diagnostic card.
Diagnostic card.
Two samples of galena: at the top - cuboctahedra from the Seravezza mine of Lucca; Below - the cubic shape in the dolomite (Joplin, Missouri)
Pb S (lead sulphide)
Cubic amount of cubic zirconia
Hardness 2,5-3
Specific weight 7,2-7,6
Cleavage is perfect
Semi-fracture fracture
Color lead-gray
Color in powder blackish gray
Glitter metal
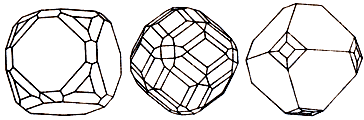
 Galena (lead gloss), - lead sulphide. Strong metallic luster. Opaque. The color is lead-gray, sometimes with a blue or reddish tinge. The line is dark gray to black. Fracture stepped. It's fragile. Cleavage is very perfect. Occurs in hydrothermal ore deposits in association with sphalerite, in stratoform deposits in the form of deposits or disseminated ores. Crystals (cubic syngony) are represented by cubes, octahedra, or combinations thereof.
Galena (lead gloss), - lead sulphide. Strong metallic luster. Opaque. The color is lead-gray, sometimes with a blue or reddish tinge. The line is dark gray to black. Fracture stepped. It's fragile. Cleavage is very perfect. Occurs in hydrothermal ore deposits in association with sphalerite, in stratoform deposits in the form of deposits or disseminated ores. Crystals (cubic syngony) are represented by cubes, octahedra, or combinations thereof.
Usually in continuous dense masses, from coarse to fine-grained. The most important and most common lead ore. The main extraction of silver is also associated with its associated extraction from galena.
Crystallizes in cubic forms, with a characteristic lead-black color. Opaque, with a metallic sheen, perfect cleavage along the edges of the cube. More often there are masses of dense or granular addition. They are distinguished by a strong shine on the surface of a fresh break, eventually blackening.
Diagnostic signs.
The mineral is soft, heavy, brittle. Quite easily melts, while a yellow lead gland (lead oxide) is formed. In hydrochloric acid (in warmth) it dissolves with the subsequent liberation of hydrogen sulphide, which has a characteristic smell of rotten egg.
Origin.
The deposits of galena are generally of magmatic origin. Formed in hydrothermal veins formed as a result of the consolidation of residual solutions, which are formed during the formation of rocks such as granites and pegmatites. In these cases, the mineral is in association with sphalerite, argentite, quartz and fluorite. Galena, in addition, can also form in sedimentary conditions. It is believed that such deposits are formed due to the concentration of minerals originally dispersed in the rock. This origin can have large deposits of galena in limestone and dolomite.

Deposits and applications.
Very rich deposits in America represent diffuse mineralization. They are in three states - Missouri, Oklahoma, Kansas. This area is called Tri-State ("three states"), and the most famous center in it is Joplin. Australian (Broken Hill), English (Cumberland), Mexican (Eulalia) and German (Andreasberg and Freiberg) deposits are also widely known. In Italy, there are deposits developed over many centuries - these are the Reibl (Carnic Alps), the Mountain Dossin (foothills of the Alps in Lombardy), Monteponi and Montevecchio (Sardinia). Galena is the main ore mineral of lead. Of some silver-containing galenics, silver is recovered as a by-product.


Galena in the concretion of phosphorite. The city of Kamenets-Podolsky, Zap. Ukraine. Photo: © А.А. Evseev.

Galena in concretion of phosphorite. Podol, Ukraine (CIS). Photo: © А.А. Evseev.

Galena from amazonite pegmatites. Flat city, Cave, Kola Peninsula, Russia (CIS). Photo: © А.А. Evseev.

Galena. The aggregate, extruded during tectonic movements of the lithosphere into the drusen cavity
Through the hole between the crystals of quartz. Berezovsk, Wed. Ural,, Russia (CIS). Photo: © А.А. Evseev.
Lead is a carcinogen and teratogen for the human body. It is vital, because it affects the synthesis of protein, the energy balance of the cell and its genetic apparatus, participates in metabolic processes of bone tissue and gonads. Normally, in the bones (cartilaginous and soft tissues) the lead content is 20 mg / kg, in the liver - 1 mg / kg, kidneys - 0.8 mg / kg, the brain - 0.1 mg / kg. In men (gonads) lead content in the body is higher than in women.
The human body contains 2 mg of lead. In the gastrointestinal tract, 5-10% (and up to 50%) are absorbed. The degree depends on the solubility of its compounds. A large amount of lead can enter the body with inhaled air (up to 70% of the aerosol containing lead is deposited in the lungs). At high concentrations of tetraethyl lead, there is a risk of penetration through the skin. Lead is excreted from the body with feces (80-90%), and a smaller part with urine.
Lead deficiency in the experiment reduces the growth of animals, disrupts iron metabolism, changes the effect of certain enzymes and the concentration of individual metabolites in the liver associated with iron status. Lead increases growth and improves hematocrit and hemoglobin concentration in iron deficiency in rats, this effect was the result of the pharmacological action of lead. The mechanism by which lead affects iron metabolism is a catalyst. Lead is necessary: with reduced hematocrit and hemoglobin, with iron deficiency conditions.
For all regions of Ukraine and Russia (CIS) lead is anthropogenic toxic element from the group of heavy metals. This is due to high pollution and exhaust emissions of motor vehicles running on leaded gasoline. From 5% to 30% of the population in cities suffer from an excess of lead. In acute lead intoxication, neurologic symptoms, lead encephalopathy, lead colic, nausea, constipation, pain throughout the body, decreased heart rate and increased blood pressure are noted. In chronic intoxication, there is increased excitability, hyperactivity (impaired concentration), depression, decreased IQ (index of intelligence evaluation), hypertension, peripheral neuropathy, loss or decrease in appetite, stomach pain, anemia, nephropathy, lead fringe, muscle dystrophy Hands, etc.
Manifestations of excess lead: increased excitability, weakness, fatigue, memory loss; headache; Lesions of the peripheral nervous system (pain in the limbs); The appearance of a leaden margin on the gum; Dental caries, arthropathy, bone system diseases, increased blood pressure, atherosclerosis, abdominal pain (lead colic), spastic constipation; Depletion, weight loss, impaired porphyrin metabolism (urobilinogen, coproporphyrin), nephropathy, progressive renal failure impairment of sperm motility and ability to fertilize, decreased potency, reticulocytosis, increased number of red blood cells with basophilic granularity, anemia, reduced resistance to infections In children), the development of the syndrome of Saturnism, a decrease in the content of calcium, zinc, selenium in the body.
- At a concentration of lead in children in the blood of 10-20 mcg / 100 ml, developmental disorders are noted.
- At an appropriate concentration of 20 to 40 μg / 100 ml, a decrease in IQ is observed.
- At lead concentration in blood in adults and children from 40 to 60 mcg / 100 ml anemia and peripheral neuropathy are noted.
- At a concentration of lead in the blood in adults from 60-80 mkg / 100 ml, chronic nephropathy is observed.
- At an appropriate concentration of 80-100 μg / 100 ml acute nephropathy is noted.
- At a concentration of 100-120 μg / 100 ml - encephalopathy.
The toxic effect of lead is due to the ability to form bonds with a large number of anion - ligands, which include sulfhydryl groups, cysteine derivatives, imidazole and carboxyl groups, phosphates. As a result of the binding of anhydrides with lead, synthesis of proteins and the activity of enzymes, for example, ATPase, are suppressed. Lead interferes with the synthesis of heme and globin, interfering with the porphyrin exchange, induces defects in erythrocyte membranes. Against the backdrop of iron, calcium, phosphorus, magnesium and zinc deficiency, the body's ability to absorb lead increases. Sulfur-containing amino acids, vitamins A, C, E, group B, folic acid, nicotinamide, calcium, magnesium, zinc, iron, chromium, phosphorus, selenium help to reduce the level of lead in the body.
ADR 1
The bomb that explodes
They can be characterized by a number of properties and effects, such as: critical mass; Scatter of fragments; Intensive fire / heat flow; bright flash; Loud noise or smoke.
Sensitivity to shocks and / or impacts and / or heat
Use the shelter, while keeping a safe distance from windows
Orange sign, the image of a bomb in the explosion
ADR 6.1
Toxic substances (poison)
Risk of poisoning by inhalation, in contact with skin or if swallowed. Dangerous to aquatic environment or sewer system
Use a mask for emergency leaving the vehicle
White diamond, ADR number, black skull and crossbones
ADR 5.1
Substances that are oxidized
Risk of violent reaction, ignition or explosion if exposed to flammable or flammable substances
Do not allow the formation of a mixture of cargo with flammable or combustible substances (eg sawdust)
Yellow diamond, ADR number, black flame above the circle
ADR 4.1

Highly flammable solids , self-reactive substances and solid desensitized explosives
Risk of fire. Flammable or combustible substances can ignite from sparks or flames. May contain self-reactive substances capable of exothermic decomposition in the case of heating, contact with other substances (such as: acids, heavy metal compounds or amines), friction or impact.
This can lead to the emission of harmful or flammable gases or vapor or spontaneous combustion. Containers can explode when heated (over-dangerous - practically do not burn).
Risk of explosion of desensitized explosives after loss of desensitizer
Seven vertical red stripes on a white background, equal in number, ADR number, black flame
ADR 8
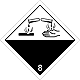
Corrosive (corrosive) substances
Risk of burns from skin corrosion. They can react violently with each other (components), with water and other substances. The substance that spilled / crumbled can emit a corrosive vapor.
Dangerous to aquatic environment or sewer system
White upper half of diamond, black - lower, equal, ADR number, test tubes, hands
| The name of a cargo that is particularly dangerous for transportation | room
UN |
Class
ADR |
| LEAD ACID HUMIDIFIED with a mass fraction of water or a mixture of alcohol and water of at least 20% | 0129 | 1 |
| LEAD OF ARSENATE | 1617 | 6.1. |
| LEAD ARSENIT | 1618 | 6.1. |
| LEAD ACETATE | 1616 | 6.1. |
| LEAD DIOXIDE | 1872 | 5.1. |
| LEAD NITRATE | 1469 | 5.1. |
| LEAD OF PERCHLORAT | 1470 | 5.1. |
| LEAD TO PERCHLORATE SOLUTION | 3408 | 5.1. |
| LEAD SOLVENT COMPOUND, NZK | 2291 | 6.1. |
| Lead stearate | 2291 | 6.1. |
| LEAD STIFNATE (LEAD TRINITRORESORCINATE) HYDROGEN with a mass fraction of water or a mixture of alcohol and water of at least 20% | 0130 | 1 |
| LEAD SULFATE, which contains more than 3% free acid | 1794 | 8 |
| LEAD-PHOSPHITE DOUBLE-REPLACED | 2989 | 4.1 |
| LEAD OF CYANID | 1620 | 6.1. |
Poisonous and radioactive dangerous stones and minerals
** - poisonous stones and minerals (mandatory check in the chemical laboratory + explicit indication of toxicity)
** - radioactive stones and minerals (mandatory check on the standard dosimeter + ban on open sales in case of radioactivity exceeding 24 milli / g / h + additional measures of population protection)
Catalog of minerals and semi-precious stones of the world by groups
** - poisonous stones and minerals
** - radioactive stones and minerals


Comments
Commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.