Native elements: Copper
 Diagnostic card.
Diagnostic card.
Cu
Cubic amount of cubic zirconia
Hardness 3,5-4,5
Specific weight 8.93
Cleavage is absent
Fracture scaly
Color copper-red
Powder Color Red Metallic
Glitter metal
Color is usually called all heavy metals, except iron, manganese and chromium. Their main representatives are copper, lead and zinc. Copper is native. The gloss is metallic, opaque. The copper-red color with brown turgidity is often covered with green powdery crusts. The dash is copper-red. The fracture is hooked. Quite forging. Cleavage is absent. Occurs in the oxidation zone of sulfide deposits of copper and in the cavities of basaltic lavas. Crystals (cubic syngony) are usually highly distorted, more often dendrites, dense nodules. Location: Lake District. Upper (USA), Zaire, Spain, the CIS. Dendrites are frequent.
Usually it is a dense mass, but also very typical of dendritic or wire form. Crystals in the form of cubes and octahedra are extremely rare. The surface is matte, but when it is fresh, it has a metallic sheen and a characteristic copper-red color. It easily undergoes changes. Copper (English Copper, French Cuivre, German Kupfer) is one of the first metals that man began to use for technical purposes.
Chemical composition. Native copper usually does not contain impurities or contains minor impurities of silver, bismuth, iron, sometimes up to 2-3% of gold (in the form of a solid solution, golden copper). Form of crystalline precipitates. Most often, solid masses, dendrites, filamentous, mossy, wire aggregates. The face-centered cubic lattice. Class of symmetry. Hexaoctahedral - m3m. Cleavage. Absent.
Diagnostic signs.
If there are doubts, they can be resolved by acting on the sample with nitric acid and moistening the solution with platinum wire. Under the action of the flame, it will turn into an intense green color. When adding ammonia solution, it is dyed blue. Easily melts and turns black. Behavior in acids. Easily soluble, with the addition of ammonia, the solution turns into deep blue.
Origin.
Copper deposits occur in ultrabasic rocks, consisting of minerals rich in iron and magnesium. They are also formed as a result of certain chemical processes in sulphide deposits. They are also found in the emptiness of basalts and conglomerates.
Mine and application.
Beautiful crystals are known in the USA (Lake Superior, Lake Arizona and New Jersey), in Germany and Finland. Native copper does not represent an industrial value. This metal, very important for the economy, is extracted from copper-bearing minerals.






Copper. Kondopoga district, Karelia, Russia. Photo: © А.А. Evseev.


Copper. Kivino Peninsula, Lake. Upper, Michigan, USA. Photo: © А.А. Evseev.

Copper. Rubik deposit, Albania. ~ 8 cm. Photo: © А.А. Evseev.

Copper. Medny Island, Commander Islands, Russia. About 10 cm. Photo: © А.А. Evseev.
In the body, copper participates in the process of breathing tissues, in the processes of anabolism (the synthesis of new structures and substances), the synthesis of hemoglobin and other ironporphyrins, skin pigments, hair, eyes, affects the functioning of the endocrine glands. In the gastrointestinal tract, up to 95% of the copper that is ingested (in the stomach as much as possible) is absorbed, in the duodenum, in the jejunum and in the ileum. Better digestible bivalent copper. Copper penetrates into all cells, tissues and organs. The maximum concentration of copper is found in the liver, kidneys, brain, blood, but copper can be found in other organs and tissues.
Copper plays an important role in the processes of biosynthesis of heme and, accordingly, hemoglobin. Therefore, its insufficiency, as well as iron, can lead to anemia. Deficiency of copper can lead to the formation of an aneurysm of the aorta and vessels of the brain. For the same reason, copper deficiency leads to demineralization of bone tissue and osteoporosis. Copper is involved in the formation of myelin sheaths of nerves (herpes, "insulators" of nerves and electric current flowing through them), the degeneration of which leads to multiple sclerosis and other disorders of the nervous system. Cobalt (in moderate doses) increases the absorption of copper. Better digestible bivalent copper.
The copper content decreases with diabetes mellitus. Its content also decreases with emotional stress, psychasthenia, epilepsy, so it seems possible to treat nervous and mental diseases with drugs and plants containing copper (psychiatry). The content of copper increases with epilepsy, hepatitis, cirrhosis, anemia, leukemia and infectious diseases (scarlet fever, diphtheria, tuberculosis, meningitis). There is a relationship between the level of copper in the blood and the increase in body temperature as a result of inflammation. Significantly, an increase in the copper content is comparable to the definition of ESR.
Copper is necessary: with hyperlipidemia, osteoporosis, anemia, to strengthen hair, normal functioning of the nervous system and joints, with lung diseases, any inflammation, gangrene, diabetes and endarteritis. Copper can inhibit the absorption by the body of iron, cobalt, zinc, molybdenum, vitamin A. Oral contraceptives, hormonal drugs, cortisone drugs contribute to the removal of copper from the body. This microelement is involved in the formation of connective tissue proteins - collagen and elastin, which are components of bone and cartilage tissue, skin, lungs, blood vessel walls.
One of the early signs of copper deficiency is osteoporosis (copper plays a role in the formation of collagen - from protein that forms bone tissue, skin and connective tissue), graying and baldness (follicles). Copper deficiency can cause growth retardation, anemia, depigmentation of hair (graying) and alopecia, weakness, decreased respiratory function, skin ulcers, loss of appetite and weight loss, cardiac atrophy, decreased hemoglobin and red blood cell count.
Manifestations of excess copper: functional disorders of the nervous system (memory impairment, depression, insomnia); Allergic dermatoses, atherosclerosis, atherosclerosis, liver and kidney function, hemolysis of erythrocytes, hemoglobin in urine, anemia, liver damage with cirrhosis and secondary brain damage (psychiatry-galutsinatsii) associated with hereditary (congenital, DNA) violation of copper and protein metabolism Wilson-Konovalov - is associated with the accumulation of copper in the liver and other tissues, the "copper eye", the rim of the pupil).
The content of copper in the liver increases with cirrhosis (serous tissue), a high level is observed with biliary cirrhosis and atresia of the biliary tract. It is recommended the appointment of chelating agents, and not the limitation of copper. Excessive consumption of copper leads to the deposition of copper in the brain tissue (brain intoxication - psychiatry), skin, liver, pancreas and myocardium (electroshock therapy).
ADR 5.1
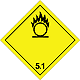
Substances that are oxidized
Risk of violent reaction, ignition or explosion if exposed to flammable or flammable substances
Do not allow the formation of a mixture of cargo with flammable or combustible substances (eg sawdust)
Yellow diamond, ADR number, black flame above the circle
ADR 6.1
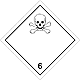
Toxic substances (poison)
Risk of poisoning by inhalation, in contact with skin or if swallowed. Dangerous to aquatic environment or sewer system
Use a mask for emergency leaving the vehicle
White diamond, ADR number, black skull and crossbones
ADR 8
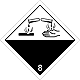
Corrosive (corrosive) substances
Risk of burns from skin corrosion. They can react violently with each other (components), with water and other substances. The substance that spilled / crumbled can emit a corrosive vapor.
Dangerous to aquatic environment or sewer system
White upper half of diamond, black - lower, equal, ADR number, test tubes, hands
ADR 9
Other dangerous substances and articles
Risk of burns. Risk of fire. Risk of explosion.
Dangerous to aquatic environment or sewer system
Seven vertical black stripes on white background - top, white - lower half of diamond, ADR number
| The name of a cargo that is particularly dangerous for transportation | room
UN |
Class
ADR |
| Copper (II) nitrate, aqueous solution, non-oxidizing | 3082 | 9 |
| Copper (II) sulfate | 3077 | 9 |
| Copper (III) Chlorate | 2721 | 5.1. |
| Copper (II) bromide | 1759 | 8 |
| Copper (II) oxychloride | 2775 | 6.1. |
| Copper (II) chloride, aqueous solution | 3082 | 9 |
| Copper (ІІ) chloride, aqueous solution, corrosive | 3264 | 8 |
| MEDI ARSENIT | 1586 | 6.1. |
| Copper Acetoarsenite | 1585 | 6.1. |
| Copper is a hemioxide. Not subject to the Regulations on the Transport of Dangerous Goods | - | - |
| Copper Dichromate | 3087 | 5.1. |
| Copper oxide. Not subject to the Regulations on the Transport of Dangerous Goods | - | - |
| COPPER CHLORIDE | 2802 | 8 |
| COPPER CYANID | 1587 | 6.1. |
| Copper dichromate Copper dichromate | 3087 | 5.1. |
| Copper chloride COPPER CHLORIDE | 2802 | 8 |
| MEDIETYLENEDIAMINE SOLUTION | 1761 | 8 |
Poisonous and radioactive dangerous stones and minerals
** - poisonous stones and minerals (mandatory check in the chemical laboratory + explicit indication of toxicity)
** - radioactive stones and minerals (mandatory check on the standard dosimeter + ban on open sales in case of radioactivity exceeding 24 milli / g / h + additional measures of population protection)
Catalog of minerals and semi-precious stones of the world by groups
** - poisonous stones and minerals
** - radioactive stones and minerals


Comments
When commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.