Oxides and hydroxides: Wolframite. Hubnerite
 Diagnostic card.
Diagnostic card.
On the picture. A sample of ferberite (Panasqueira, Portugal), the most iron-rich mineral from the wolframite group.
(Fe, Mn) WO 4
Singonia monoclinic
Hardness 5-5,5
Specific weight 7.14-7.54
Cleavage is perfect
Crack irregular
Color reddish brown, black-brown
Color in powder reddish-brown, black
Gloss semi-metallic, resin
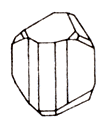
Wolframite is a tungstate of iron and manganese. The gloss is metallic, opaque. The color is black or dark brown. The line is black to brown. The fracture is uneven, brittle. Cleavage is perfect. It is formed in quartz dwellings and greisens. Crystals (monoclinic syngony) prismatic habit, aggregates - solid, dense. Along with scheelite, wolframite is the most important tungsten ore. Deposits: in China, Burma, USA, Bolivia, Portugal. Opaque mineral of red or black color, which is the most important raw material for the production of tungsten.
The wolframite crystallizes in the monoclinic syngony. The crystals are thickly tabular, the faces are shaded so much that the crystals themselves sometimes appear to consist of plates. The color is reddish-brown, to black-brown. It is often found in the form of opaque granular masses. The powder has a reddish-brown color. Gloss is semimetallic, on the surfaces of cleavage resin. It does not dissolve in acids and practically does not liquefy oxidation. It is a very heavy, hard and brittle mineral. The term "wolframite" is used to designate an intermediate mixture in the series isomorphous from manganese tungstate (gubnerite) to iron tungstate (ferberite).
The chemical composition is variable; WO4 content to 76.6%; By the number of FeWO4 molecules (in%) differ: ferberite (100-80), actually wolframite (80-20), gubnerite (20-0); Admixtures of iron, calcium, magnesium, niobium, copper, etc. are noted. Transparency is opaque (ferberite) to translucent and transparent in thin cleavages (gybnerite).
Singonia is a monoclinic, prismatic type of symmetry. Huberite is characterized by prismatic elongated crystals, for wolframite - short-prismatic, ferberite stretched along [010] and flattened by [100]. The main simple forms are: [100], [110], [102], [001], [010] and [011]. Often there are twins according to [100] and [023]. Longitudinal shading is observed on the vertical faces of the crystals. Radial-ray, needle-like aggregates, solid massive discharges of different degree of granularity are also common. Cleavage is perfect by (010).
Diagnostic signs.
Wolframite can be confused with some sulfides and minerals of the columbite group. The latter differ in cleavage, which is fairly well seen, and by the fact that well-crystallized columbite is almost always found in pegmatites. From sulphides differs when exposed to potassium bisulphate and then treated with a solution of sulfuric acid with pieces of zinc. This produces a magnificent blue color. Color of the dash: in ferberite - dark brown, in gyubnerita - yellow, yellow-brown
Origin.
Wolframite is a typical mineral of pneumatolite genesis (the result of exposure to residual magmatic solutions after crystallization of granites). Fluids (steam-gas mixtures) are introduced into the cracks of solidifying surrounding rocks and form quartz veins in which wolframite crystallizes. Due to their high specific gravity and weathering resistance, wolframite also accumulates in sedimentary alluvial deposits, which are formed from the products of rock destruction, transported by river streams and accumulating in alluvium.
Place of Birth.
Large deposits are located in the south of China (the Nan-Lin mountain chain), Australia (Queensland), Bolivia (La Paz region), Canada, the USA (California and Colorado), Malaysia and Burma. In addition, wolframite is found in the Cornwall Peninsula and in Cumberland (Great Britain), in the Ore Mountains of Saxony and Bohemia and in Portugal.
Application.
Wolframite is the main ore on tungsten; It contains up to 60% of this metal. Used in metallurgy; Of tungsten make spirals of electric lamps, and its carbide is used for drilling and drilling tools and special steels.






Hubnerite (2-3.5 cm) on a drift of smoky quartz. Akchatau, Sev. Pribalkhash, Kazakhstan (CIS). Photo: © А.А. Evseev.
Mn WO 4 - Fe-gubnerite (wolframite) - tungsten ore. Color - reddish-brown, black-brown

Gyubnerite (black crystals up to 2 cm). Arsenievskoye deposit, Primorye, the Russian Federation (CIS). Photo: © А.А. Evseev.

Hubnerite (wolframite - black), quartz. Silverton, NY Colorado, USA. Photo: © А.А. Evseev.

Gübnerit. Mine to them. Dzhambul, Kara-Oba, the Center. Kazakhstan (Central Asia, CIS). Photo: © А.А. Evseev.
On average, about 0.001-0.015 mg of tungsten per day is absorbed into the human body from food. The digestibility of tungsten and its salts in the human gastrointestinal tract is on the average 1-10%, and weakly soluble tungstic acid - up to 20%. Accumulation of tungsten occurs in the bones and kidneys (lumps the coms and kidneys with its excess). The bone tissue contains 0.00025 mg / kg, in the blood - 0.001 mg / l of tungsten. Tungsten is excreted from the body with urine, and 75% of its radioactive isotope 185W - with feces. Molybdenum is a tungsten antagonist. On the properties of tungsten resembles molybdenum, however, unlike molybdenum, tungsten is not an essential (concentrating) element. With the accumulation of tungsten salts, the level of uric acid decreases and the levels of xanthine and hypoxanthin increase, which is important for gout.
Chronic entry of tungsten dust into the body can lead to the development of a clinical syndrome - "heavy metal disease" or pneumoconiosis. The most common symptoms are cough, breathing disorder, atopic asthma and changes in the lungs. The manifestation of symptoms decreases after a change of place of work or a long rest, due to the termination of contact with the metal. In severe cases, with late diagnosis of the disease, the pathology of the pulmonary heart, lung fibrosis and emphysema can develop.
WOLFRAM, W (Latin Wolframium, * a. Tungsten, Wolfram, tungstene, and tungsteno), - a chemical element of group VI of the Mendeleyev periodic system, atomic number 74, atomic mass 183.85. Natural tungsten consists of a mixture of five stable isotopes 180W (0.135%), 182W (26.41%), 183W (14.4%), 184W (30.64%), 186W (28.41%). Opened and isolated in the form of tungsten anhydride in 1781 Swedish chemist K. Scheele. In 1783, the chemists of D'Olouard isolated WO3 from wolframite and, recovering it with carbon, received a pure metal called tungsten. A chemical element, a refractory metal of light gray color, used in various alloys, for the manufacture of filaments in electric bulbs, as well as in the production of paints.
Tungsten is a heavy metal, light gray, refractory. It crystallizes in a body-centered cubic lattice with a period a = 0.31647 nm (3.1647 E). Density 19300 kg / m3, t melting 3410 ± 20 o C (after carbon the most refractory element), boiling point 5930 o C. Thermal conductivity [W / (m * K)] 129.89 (20 o C); 108.94 (1300 ° C). Specific electrical resistance (Ohm * m) 5.5 * 10-4 (20 ° C); 90.4 * 10-4 (2700 ° C). The temperature coefficient of electrical resistance (0-170 o C) 5,1 * 10-3 K-1. Important properties of tungsten: high electron emission at incandescence of metal (mA / m2) - 1.5 * 10-6 (830 ° С); 2.3 (1630 ° C); 104 (1730 ° C); 298 * 104 (2230 ° C) and 1690 * 104 (2427 ° C); Large power radiated by metal surface energy at high temperatures (W / m2): 0,9 * 104 (800 o C); 18.0 * 104 (1600 ° C); 64.0 * 104 (2200 ° C); 153.0 * 104 (2700 ° C); 245 * 104 (3030 ° C).
In compounds, the degree of oxidation of tungsten can be from +2 to +6. In higher degrees of oxidation, it has acidic properties, in the lower ones - basic. The compounds of the lower stages of tungsten oxidation are relatively unstable. The most characteristic and stable are tungsten compounds with an oxidation state of +6. Tungsten has a great propensity to complex formation. Metal tungsten under normal conditions is chemically very stable. With oxygen, it begins to react at temperatures above 400 ° C; Counteracts the action of water, but at a temperature of red heat it is easily oxidized by water vapor. Tungsten in the cold is practically not exposed to HCl, H2SO4, HNO3 and HF of any concentration, but it dissolves easily in a mixture of HNO3 and HF. In the absence of oxygen, tungsten does not dissolve in alkali and ammonia. The most important of the tungsten compounds are WO3 trioxide, tungstic acid H2O4 and its salts are tungstates.
Tungsten is not widely distributed in nature; The content in the earth's crust is 1 * 10-4% (by weight). In the free state does not occur. Forms its own minerals (tungstates Ca, Fe, Mn, sometimes Pb, Zn, rarely oxides of WO3, H2O4, even less often sulfides WS2) or enters in the form of isomorphic impurities in other minerals, mainly in minerals Mo, Ti, and also in some silicates Mica, feldspars).
The most important minerals of tungsten are wolframite and scheelite, which can be formed and accumulated to the level of industrial concentrations in skarn, greisen and hydrothermal processes. In natural mineral parageneses, tungsten is often associated with Si, Mo, Sn, Be, Ta, F, more rarely - with Au, Sb, Hg. The forms of migration of tungsten in high-temperature ore-bearing solutions are mainly represented by hydroxy- and hydroxyfluoride complexes. On the main genetic types of tungsten deposits and enrichment schemes, see Art. Tungsten ores. The production of pure metal from tungsten concentrates is carried out in three stages: the chemical isolation of pure tungstic acid or its salts; Reduction of WO3 to metal powder; The transformation of the powder into a metal.
The main area of application of tungsten is the production of steels (about 85% of production). Pure tungsten is used for the production of filaments of electric lamps, coils of heaters in electric furnaces, electrodes, various parts for high-vacuum and X-ray devices, and atomic-hydrogen welding. On the materials: http://www.mining-enc.ru
ADR 2.1


Highly flammable gases
Risk of fire. Risk of explosion. Can be under pressure. Risk of suffocation. May cause burns and / or frostbite. Capacities can explode when heated (over-dangerous - almost do not burn)
Use shelter. Avoid low areas of the surface (pits, lowlands, trenches)
Red diamond, ADR number, black or white flame
ADR 2.2


Gas container Non-flammable, non-toxic gases.
Risk of suffocation. Can be under pressure. May cause frostbite (similar to a burn - pallor, blisters, black gas gangrene - creaking). Capacities can explode when heated (highly dangerous - explosion from a spark, flame, matches, practically do not burn)
Use shelter. Avoid low areas of the surface (pits, lowlands, trenches)
Green diamond, ADR number, black or white gas cylinder (such as "balloon", "thermos")
ADR 2.3
Toxic gases . Skull and crossbones
Danger of poisoning. Can be under pressure. May cause burns and / or frostbite. Capacities can explode when heated (highly dangerous - instantaneous gas spreading around the neighborhood)
Use the mask for emergency leaving the vehicle. Use shelter. Avoid low areas of the surface (pits, lowlands, trenches)
White diamond, ADR number, black skull and crossbones
| The name of a cargo that is particularly dangerous for transportation | room
UN |
Class
ADR |
| TUNGSTEN HEXAFLUORIDE | 2196 | 2 |
- Ghetchellit - "New Almaden blend" - arsenide and antimony sulfide (modern sulfosol)
- Antimony is a toxic metal (semimetal) , widely used in metallurgy, medicine and engineering
- Zirconium - a rare and undiscovered metal and the most dangerous precious stone in oxide and salt
- Gold - yellow dangerous and poisonous metal of modern accurate digital and cable technologies
- Sulfur is a golden-yellow toxic substance and a sign of active volcanic activity
- Cadmium is an undisputed toxic silvery metal unknown to a wide range of people
- Lead - a poisonous gray imitator of metallic silver and toxic metal blende
- Arsenic is a classic poison of medieval and modern poisoners and medicine in medicine
Poisonous and radioactive dangerous stones and minerals
** - poisonous stones and minerals (mandatory check in the chemical laboratory + explicit indication of toxicity)
** - radioactive stones and minerals (mandatory check on the standard dosimeter + ban on open sales in case of radioactivity exceeding 24 milli / g / h + additional measures of population protection)
Catalog of minerals and semi-precious stones of the world by groups
** - poisonous stones and minerals
** - radioactive stones and minerals


Comments
Commenting on, remember that the content and tone of your message can hurt the feelings of real people, show respect and tolerance to your interlocutors even if you do not share their opinion, your behavior in the conditions of freedom of expression and anonymity provided by the Internet, changes Not only virtual, but also the real world. All comments are hidden from the index, spam is controlled.